14. Cell Division and the Cell Cycle (1,218)
- lscole
- Apr 28, 2025
- 5 min read
Updated: May 2
Every day, your body replaces hundreds of billions of cells.
Each one must copy over three billion letters of DNA and divide its contents with extraordinary precision.
And it must do so at the right time—no sooner, no later.
The importance and rate of this process vary dramatically. Some tissues--like bone marrow, the lining of the gut, the skin, and hair follicles--are constantly turning over. Their cells divide continuously to replace those lost to wear. Others divide rarely or not at all.
There is also a developmental dimension. During embryonic development, cells divide at an fast pace. By birth, the human body contains about one trillion cells, meaning at least that many divisions have occurred to build the body from a single fertilized egg.
And after birth, division is more frequent during infancy and childhood than in adulthood.
Cell growth and renewal depends on a single recurring process: the cell cycle.
The cell cycle is not just a sequence of phases--it’s a progression of commitments. At each stage, the cell makes a decision that commits it to the next step, often irreversibly once key checkpoints are passed.
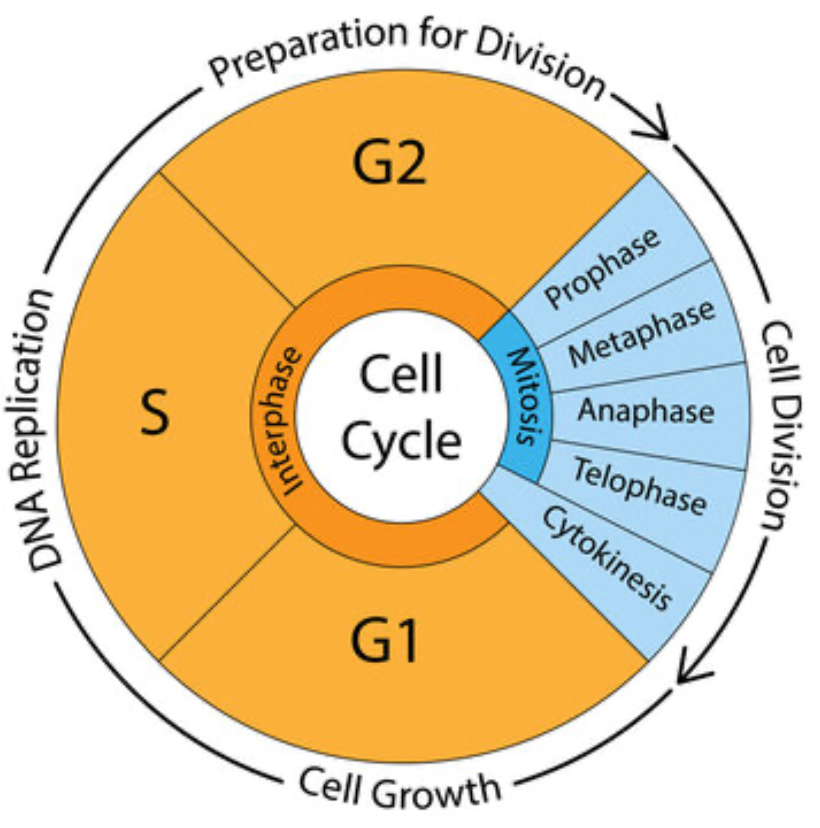
The cell cycle
These phases are not just steps—they are decisions.
Then lightly tag each phase:
G1: whether to divide
S: commit to copying the genome
G2: confirm everything is ready
M: divide
The cell cycle has one primary purpose: to prepare for and then to execute genome replication and cell division.
For many human cells the cycle takes roughly 24 hours, though the timing varies widely. Those 24 or so hours are divided into four phases in the following order:
G1 for "growth 1" -- the cell decides whether to divide
S for "synthesis" -- the cell commits to copying the genome
G2 for "growth 2"-- the cell confirms everything is ready
M for "mitosis"-- the cell divides
Cells that aren't actively dividing but just doing their jobs are often described as being in G0 ("G zero"). G0 is not part of the cell cycle. But a cell in G0 can enter the cell cycle at G1 if growth signals from the surrounding tissue reach it.
Of the four phases of the actual cell cycle, the S phase and M phase are primary--they deliver the two outputs of the cell cycle: genome replication and cell division.
The other two phases, G1 and G2, are focused on, respectively, committing to and preparing for S phase, and confirming that the cell is ready to divide in M phase.
S phase is one of the most dangerous tasks a cell performs. A single error can propagate through all future cells.
There are several distinct and highly choreographed steps in M phase. We'll discuss these in the next chapter.
As mentioned, the G1 and G2 phases play critical but supporting roles.
During G1 the cell grows and assembles the machinery for DNA replication. At the end of G1 there is a cell cycle checkpoint; the G1/S checkpoint. At this checkpoint the cell determines whether it should commit to another round of division.
Following S phase, the cell enters G2 where it prepares for division, or M phase. To do so it produces additional organelles and other cellular components to fill the two daughter cells.
Another checkpoint occurs at the end of G2: the G2/M checkpoint. Here the cell checks that the DNA copied during S phase is complete and free of major damage and that the cell is ready for mitosis.
One more thing: After M phase there is a short phase called cytokinesis during which the cell pinches itself off completely at its midpoint to become the two new daughter cells.
After cytokinesis, the daughter cell enters G1. From there it may proceed through another round of the cell cycle or exit into G0, where it can carry out its specialized function(s) without dividing.
Regulating the cell cycle
The cell doesn’t use a central clock. It keeps time through a sequence of molecular triggers.
At each phase of the cell cycle, new sets of genes are turned on and others turned off to accomplish the tasks of that phase.
This turning on and off of the appropriate genes is spearheaded by members of two protein families that work as partners: the CDK (cyclin-dependent kinases) family of enzymes and the cyclin family of regulatory proteins.
A CDK is a class of enzyme called a kinase that uses an ATP molecule to transfer a phosphate to a specific amino acid on another protein. The placement must be precise.
Kinase-attached phosphate groups change protein behavior. They can serve to identify, or flag, a protein as a target for an action by another protein. They can also change the conformation (shape) of the protein to activate it, deactivate it, or alter its function.
Kinase-attached phosphates are powerful molecular switches with different purposes depending on the protein, where it is attached, and the cellular context.
Members of the CDK family are present in the nucleus all the time but remain inactive unless bound to a partner cyclin. They're like machines wandering around waiting for the right key.
The expression of different cyclins drives different phases of the cell cycle. When a particular cyclin is expressed, it binds to a specific partner CDK and switches it on.
Being a kinase, the activated CDK then phosphorylates many other target proteins, triggering the events required for that phase of the cycle.
As one phase ends, its associated cyclin is destroyed and replaced by another, activating a new set of CDKs and moving the cell forward through the cycle.
One example will give you the flavor.
We'll begin at the start of G1 phase. Cyclin D activates two partner CDKs: CDK4 and CDK6.
As the cell approaches the G1/S transition, cyclin E takes over, activating CDK2, which prepares the cell to copy its genome.
During S phase, cyclin A attaches to CDK2. This drives DNA replication.
Later, in G2 and M phase, cyclin A and cyclin B activate CDK1. This carries the cell through mitosis and prepares it for cytokinesis.
No single molecule oversees this process. Instead, each step triggers the next.
The rollout of these cyclins must be precise because genome copying and cell division are two of the most dangerous things a cell does. Nearly every major failure of cell regulation, including many forms of cancer, traces back to mistakes made in replication and cell division.
And yet, this process unfolds reliably, again and again--not because a central system directs it, but because each step triggers the next.
In the next chapter, we’ll look closely at mitosis--where the coordination becomes visible as chromosomes are pulled apart with amazing precision.
(1) Scientific American, April 1, 2001, Our Bodies Replace Billions of Cells Every Day. https://www.scientificamerican.com/article/our-bodies-replace-billions-of-cells-every-day/?utm_source=chatgpt.com
(2) Bionumbers. Accessed 1-15-2025. https://bionumbers.hms.harvard.edu/bionumber.aspx?id=106413&ver=4&utm_source=chatgpt.com
Most of the time, most of your cells are just doing their jobs. They're not dividing.
Nerve cells are transmitting electrochemical signals. Heart muscle cells are contracting. Rod and cone cells in the retina are detecting this text.
But it's also true that your body is constantly renewing itself. Every day it replaces billions of cells. That’s an enormous number--but it still represents less than one percent of all your cells.

Comments