12. Flexing Your Muscle Cells (1,053)
- lscole
- Apr 22, 2025
- 4 min read
Updated: Apr 30
Flex your arm. It feels smooth, controlled, almost effortless.
But beneath that motion, billions of molecular events are unfolding--tiny proteins pulling, releasing, and resetting in rapid succession.
Meet myosin, a protein complex in skeletal muscle cells. In this chapter, we'll see how ATP enables molecular-scale myosin movements that cause skeletal muscle contractions.
A muscle contraction is not a single event--it’s the result of billions of coordinated molecular movements.
We’ll zoom in step by step--from the level of muscle cells down to individual myosin molecules.
The first level is muscle cells, but in this field, muscle cells are called muscle fibers due to their elongated shape and distinct characteristics. I'll stay aligned with the field and use the term fiber rather than cell in this chapter.
Inside these muscle fibers are numerous myofibrils--long, rod-like structures composed of repeating sarcomeres.
Within a sarcomere, thick myosin filaments and thin actin filaments are arranged so they can slide past one another. This enables them to operate as units of molecular contraction.
The coordinated movement of sarcomeres drives myofibril contraction, which in turn drives contraction of the muscle fiber--and ultimately the muscle itself.
Our ultimate focus is how ATP makes myosin move.
Muscle fibers
Skeletal muscles are composed of muscle fibers. These are highly specialized cells that don't look like the typical cell I've described so far.
Muscle fibers are extremely long--typically 1 to 5 cm long, but they can reach 30 cm, which is about a foot long.
Because they are so long, they require multiple nuclei. Each serves the region of the fiber near it.
They also contain many mitochondria due to their high energy requirement. Most human cells have hundreds of mitochondria. Muscle cells contain thousands.
Finally, muscle fibers have a modified cell membrane called a sarcolemma that allows the fiber to respond to nerve impulses instructing it to contract.

Myofibrils and sarcomeres
Muscle fibers are packed with myofibrils made of myosin and actin proteins arranged into contractile units called sarcomeres.
To understand sarcomeres, we'll need some new terms and some figures.
Sarcomeres consist of a thick filament made of long myosin molecules, and a thin filament made of two long actin molecules twisted around each other. Each myosin has a head. These are the round extensions that look like lollipops.
Thick myosin filaments are attached to the M line--a central wall made of structural proteins. Myosins extend outward in both directions from this M line.
The actin thin filaments are also permanently attached, but to two outer walls called Z lines. One thin filament extends inward from the left Z line. The other extends inward from the right Z line.
The actin thin filaments interlace with the myosin thick filaments.
At its core, a sarcomere is a simple machine: thick myosin filaments in the center and thin actin filaments anchored on either side.
The coordinated movement of sarcomeres causes myofibril contraction, which in turn causes contraction of the muscle fiber--and ultimately the muscle itself.
But why does the sarcomere contract?
Look closely at the bottom drawing in the figure. It shows a sarcomere in a relaxed state and in a contracted state. In the relaxed state, the myosin heads are prevented from binding to actin. They lie flat against the thick filament. In the contracted state, the heads stick out from the thick filament.
In the transition from the relaxed to the contracted state, the myosin heads sweep back toward the M line. Before they do, they grab actin thin filaments and pull them inward toward the M line. This pulls the two Z lines inward toward the M line.
That is a sarcomere contraction.
The interaction resembles a cyclic rowing motion, with repeated strokes pulling the actin filaments inward and thus pulling the Z lines toward the central M line. This is what makes the sarcomere contract.
Myosin and ATP
So far, we’ve described what happens. But none of this explains what drives the motion. For that, we need ATP.
Recall that when the third phosphate is removed, turning ATP into an ADP (adenosine diphosphate) and a lone P (phosphate), the result is a burst of energy. I likened the energy burst to a compressed spring being released or to a kick.
Just as a spring being released and a kick can cause movement, the transition from ATP to ADP + P also causes movement: molecular movement.
We're now ready to really go under the hood to see myosin head movement. The steps are easiest to see in the figure below.
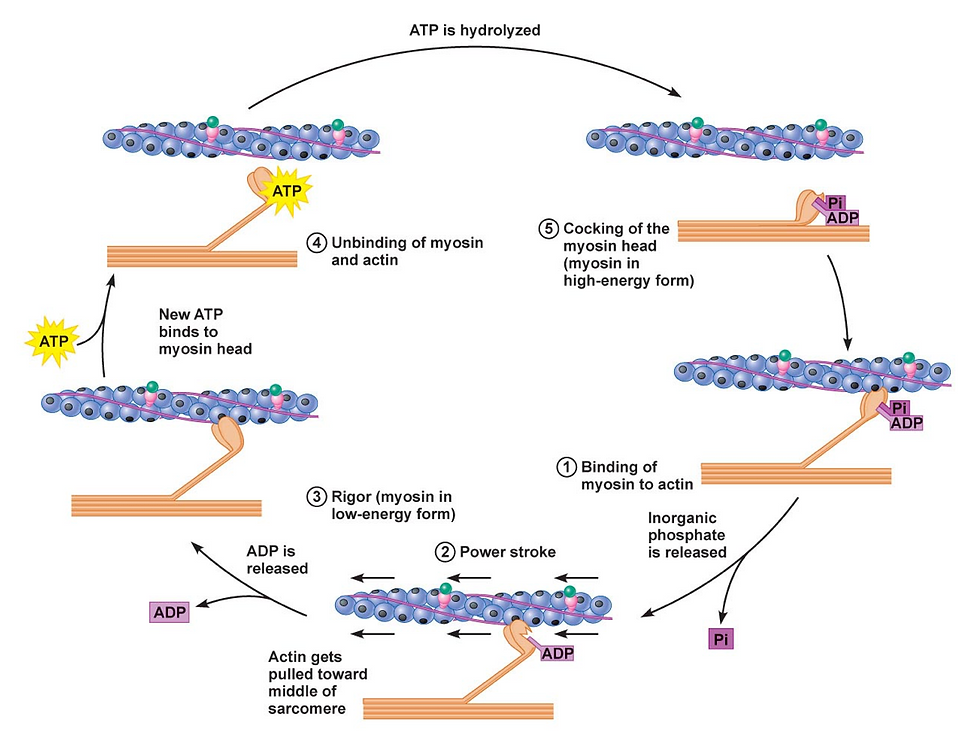
Step 1. Binding to actin. We start with the myosin head attached to the actin thin filament. In this position, ADP and P are attached to the head.
Step 2. Power stroke. Release of the P from the myosin head strengthens its binding to actin and triggers the power stroke. The myosin pulls the actin thin filament toward the center M line and causes sarcomere contraction.
Step 3. ADP release. Soon after the power stroke, the ADP molecule is released from the myosin head, leaving the ATP/ADP binding site vacant. The myosin head is still attached to actin at this point.
Step 4. ATP binding. When ATP arrives at the myosin binding site, it causes detachment of the myosin head from the actin thin filament.
Step 5. ATP cleavage (energy used). Finally, when the ATP is cleaved into ADP and P, it’s as if a spring is reset or a leg bends in preparation for the next kick.
This puts the myosin protein in position for the next power stroke. The resulting ADP and P remain attached to the myosin head and we cycle back to Step 1.
So, in the case of myosin head movement, the cleavage of ATP does not actually cause the power stroke. Instead, the energy is used to cock the myosin head so that when it is released the power stroke occurs.
Every time you move a muscle, millions of these molecular events occur--myosin heads binding, pulling, releasing, and resetting. What we experience as smooth, coordinated motion is really the sum of countless tiny mechanical actions.
This is a recurring theme: large-scale biological phenomena emerge from simple molecular processes repeated again and again.
This same principle will appear again in genome replication. Simple molecular rules repeated at scale can produce something remarkably precise.

Comments